|
N-Nitrosamine compounds, such as N-nitrosodiethanolamine (NDELA), have long been
recognized as a class of hazardous compounds.The potential cancerogenity demands
effective and reliable analysis.
N-nitrosamine compounds are found in:
- Foods including: dried meats, beer, cheese, fish, bacon, other cured meats
- Cosmetic products including: shampoos,
- Tobacco smoke, chewing tobacco
- Tanneries and plants manufacturing pesticides
- Rubber products and tires
- Air – especially in factories participating in the use or manufacture of rubber products
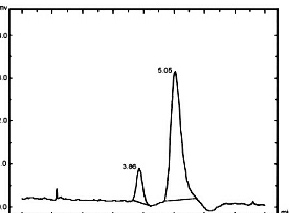
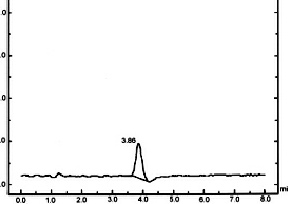
International Standards Organization approved method: ISO 10130:2009 is an analytical
method for the detection and determination of NDELA in cosmetics through HPLC using
post-column photolysis and chemical derivatization. The method is both highly sensitive
and relatively simple. By employing the Aura post-column photochemical reactor,
PHRED™, and post-column chemical derivatization unit, EPOCOD™, Aura Industries has
evaluated the ISO method and have shown that it provides quick, accurate and economically
feasible determination of NDELA and other N-nitrosamine compounds.
Apparatus, Chemicals and Chromatographic Conditions
HPLC Parameters:
Column: Waters Nova-Pak® C18 Column, 15 cm x 4.6 mm, 5 µm particles, with CJB-10
column jacket (Aura Industries, Inc.)
Temperature of column maintained with circulating water bath: 40°C
Mobile Phase: 0.02 M ammonium acetate solution. 0.5mL/min. flow rate. pH 7.6. Phosphate
buffer pH 7.6 can also be substituted.
Flow rate: 0.5 mL/min
Injection Volume: 100 µL
|