Formaldehyde or formaldehyde releasing preservatives are frequently used in cosmetic
preparations such as shampoos and skin-care products for the prevention of microbial
and fungal growth. Many countries and the European Union have applied restrictions
on the concentration of formaldehyde measured in the free form in such products (1).
Accurate determination of free formaldehyde concentration is essential for correct
labeling and classification of many products in accordance with these regulations.
In the HPLC method used here, free formaldehyde elutes as an early peak (1.83 min.)
and is chemically derivatized with the Nash reagent (2) through a post column derivatization
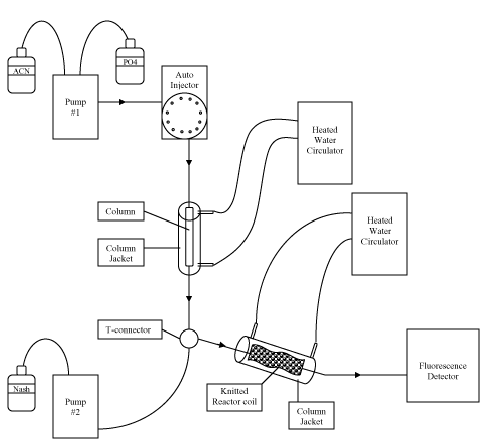
reaction. A schematic of the HPLC-post column derivatization setup is shown in Figure
1. For clarification, it is generally accepted that formaldehyde in its hydrated
form of methylene glycol and polymethylene glycol is derivatized to completion with
reagents like DNPH and Nash. It is assumed, therefore, that the term “free formaldehyde”
refers to monomeric formaldehyde, methylene glycol and polymethylene glycol (2).
This method was found to be specific for formaldehyde and gave no response with acetaldehyde,
propionaldehyde, buteraldehyde or benzaldehyde. A calibration curve was determined
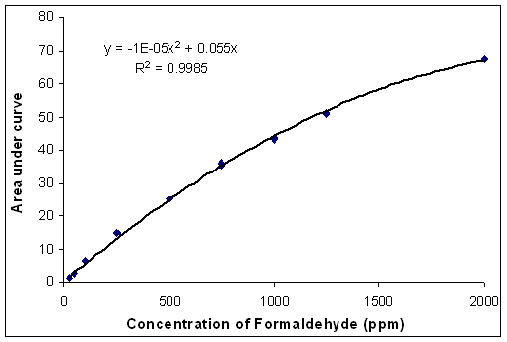
for formaldehyde (Figure 2) under the stated conditions. The calibration curve follows
a polynomial function (R2 = 0.9985). Four commercial cosmetic preservatives and five
cosmetic surfactants were assayed for formaldehyde. The concentration of formaldehyde
was calculated using the polynomial formula derived from the calibration curve (Figure
2). No detectable level of free formaldehyde was found in surfactants: Steol® CS-370,
Steol® CS-270, and Stepanol® WAT-K. The formaldehyde concentrations of the six remaining
samples ranged from 2ppm (Stepanol® WAT) to 1084ppm (Glydant® XL-1000) (Figure 4).
Dowicil™ 200 Preservative displayed an initial formaldehyde peak at 1.83 min. at
a concentration of 100ppm but also displayed a second peak (3.02 min.) which was
most likely additional formaldehyde being released more slowly from the Dowicil molecule
(Figure 4, G.). The lower limit of detection of free formaldehyde using the stated
method is 0.7 ppm (calculated as 3 times the background noise). Twelve replicate
samples of 500 ppm formaldehyde were run and the calculated percent standard deviation
of response and retention time were 1.70% and 0.34% respectively
This improved methodology for the analysis of free formaldehyde in cosmetic preparations
employs several cost effective changes to existing protocols. The use of a peristaltic
pump as part of a standard EPOCOD™ to deliver the post column chemical derivatization
agent provides a consistent flow rate that would otherwise have required an additional
HPLC pump. We have also dispensed with the need for the expensive acetonitrile solvent,
which is more commonly used for this type of procedure (2) and replaced it with methanol.
Finally, we used Poland Spring® Brand distilled water for reagent and sample preparations
rather than a much more expensive HPLC grade water.
Materials and Methods:
HPLC Parameters:
Column: Nova Pak C18, 3.6 x 150mm (Waters), Column Jacket (CJB-10) (Aura Industries,
Inc.)
Temperature: 40°C
Mobile Phase: 60:40 Methanol (MeOH):0.01M H3PO4
Flow rate: 1 ml/min.
Injection: Micromeritics 728 Autosampler – 20µL sample
Post Column Conditions:
A Masterflex® peristaltic pump was employed in conjunction with a Post Column Derivatization
kit (EPOCOD™) and 5-50 KRC(Aura Industries, Inc.)
Flow rate: 0.8 mL/min.
Temperature of post column reactor: 70°C
Derivatization Agent:
Nash Reagent: 77 g of ammonium acetate were dissolved in roughly 250mL of Poland
Spring® Brand distilled water. 1.25mL of glacial acetic acid and 1 mL of acetylacetone
were added. After vigorous mixing, the solution was transferred to a 0.5-L volumetric
flask, followed by 200 mL of water. The pH of the solution was adjusted to 5 with
glacial acetic acid, and the mixture diluted to 0.5-L the mark with water. The solution
was then filtered through an IFDA (Aura Industries, Inc.) with TFM 1.5-10 teflon
filter membrane ~1.5mm pore size (Aura Industries, Inc.).
Detection:
Applied Biosystems 980 programmable Fluorescence Detector
Emission Filter: 550nm, Excitation wavelength: 410nm
Sample preparation:
Formaldehyde (37% in water/methanol), (Sigma-Aldrich) was diluted to concentrations
ranging from 2000ppm to 1ppm with Poland Spring® Brand distilled water:methanol (85:15).
Butyraldehyde and Proprionaldehyde (Sigma-Aldrich) were diluted to 2500ppm with 85:15
water:MeOH solution r and Benzaldehye and Acetaldehyde were diluted to 1000ppm. Preservatives:
Dowicil™ 200 Preservative (Dow Chemical Midland, MI), Glydant® and Glydant® XL-1000
(Lonza Inc. Allendale, NJ), Germall® 115 (Sutton Laboratories Chatham, NJ) 0.4g of
each were dissolved in 40mL 85:15 water:MeOH solution to yield a 1.0% solution of
each. Surfactants: Steol® CS-370, Stepanol ® WAT-K and Stepanol® AM (Stepan Company,
Northfield, IL) 0.4g of each were dissolved in 40mL 85:15 water:MeOH solution to
yield a 1.0% solution of each. Surfactants: Steol® CS-270 and Stepanol ® WAT (Stepan
Company, Northfield, IL) 0.8g of each were dissolved in 40mL 85:15 water:MeOH solution
to yield a 2.0% solution of each.
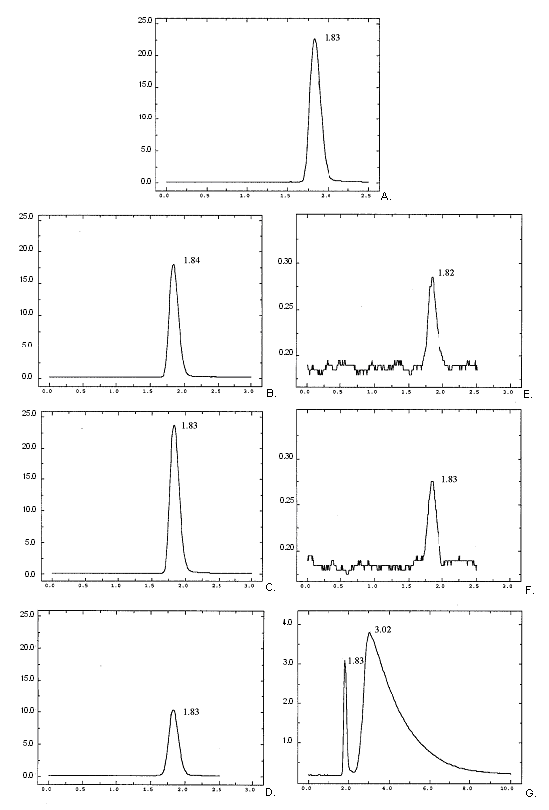
Figure 3. Chromatograms of (A) formaldehyde 1000 ppm dilution, (B) Glydant ® 10,000
ppm solution, formaldehyde concentration: 734ppm (C) Glydant® XL-1000, 10,000ppm
solution, formaldehyde concentration: 1020ppm, (D) Germall® 115 10,000ppm solution,
formaldehyde concentration: 420ppm, (E) Stepanol® AM, 10,000pm solution, formaldehyde
concentration: 2ppm, (F) Stepanol ® WAT, 20,000ppm solution, formaldehyde concentration:
2.5ppm, (G) Dowicil™ 200, 10,000ppm solution, formaldehyde concentration: 101ppm.
Retention times for each sample are shown on the graph. Note the difference of scale
on the Y axis for (E,F,G).